 Pre-IND application, to review FDA guidance documents and get answers to questions that may help enhance their researchĪfter Phase 2, to obtain guidance on the design of large Phase 3 studiesĪny time during the process, to obtain an assessment of the IND applicationĮven though FDA offers extensive technical assistance, drug developers are not required to take FDA’s suggestions. In the IND application, developers must include:Īnimal study data and toxicity (side effects that cause great harm) dataĬlinical protocols (study plans) for studies to be conductedĭrug developers are free to ask for help from FDA at any point in the drug development process, including: Phase 4 trials are carried out once the drug or device has been approved by FDA during the Post-Market Safety Monitoringĭrug developers, or sponsors, must submit an Investigational New Drug (IND) application to FDA before beginning clinical research. How the data will be reviewed and analyzedĬlinical trials follow a typical series from early, small-scale, Phase 1 studies to late-stage, large scale, Phase 3 studies. What assessments will be conducted, when, and what data will be collected How the drug will be given to patients and at what dosage Whether there will be a control group and other ways to limit research bias How many people will be part of the study Who qualifies to participate (selection criteria) Before a clinical trial begins, researchers review prior information about the drug to develop research questions and objectives. 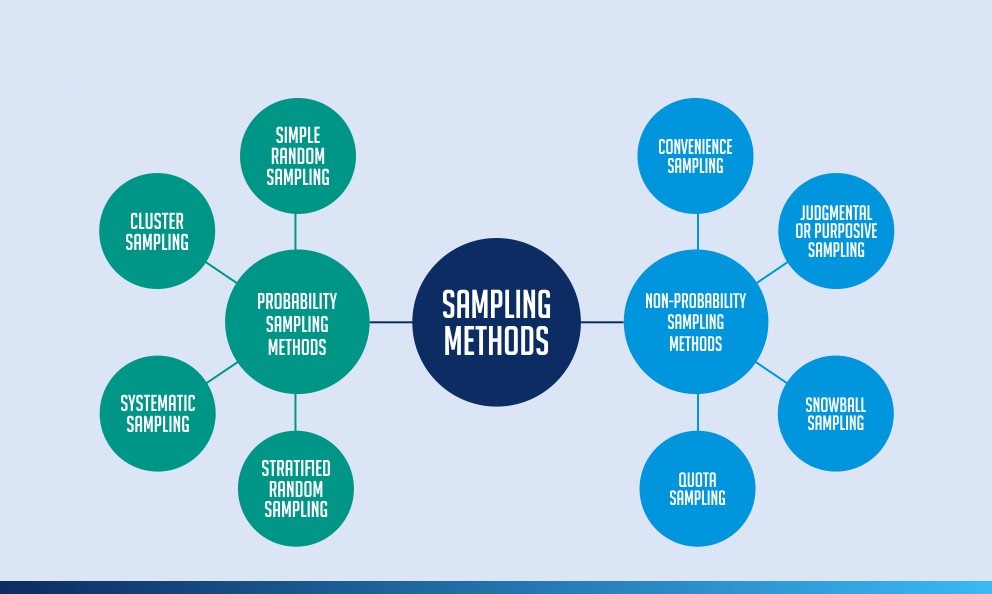
These trials follow a specific study plan, called a protocol, that is developed by the researcher or manufacturer. Researchers design clinical trials to answer specific research questions related to a medical product. On this page you will find information on: 
As the developers design the clinical study, they will consider what they want to accomplish for each of the different Clinical Research Phases and begin the Investigational New Drug Process (IND), a process they must go through before clinical research begins. “Clinical research” refers to studies, or trials, that are done in people. 
While preclinical research answers basic questions about a drug’s safety, it is not a substitute for studies of ways the drug will interact with the human body. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
